Chemokine signaling to integrin activation
Chemokines are the most powerful physiological integrin activators, capable of triggering the complete conformeric transition of the integrin heterodimer from low (bent) to high affinity (fully extended) state. Chemokines also control heterodimer lateral mobility on the plasma membrane, leading to increased valency. Complex intracellular signaling mechanisms mediate integrin affinity maturation by chemokines.
We have previously described several signaling mechanisms of integrin activation by chemokines. Specifically, we have studied the role of rho small GTPases, PKCs isoforms, PKA, PIP3K, PLD1 and PIP5K1C (refs.). Very recently, we have demonstrated the critical role of JAK PTKs in the hierarchical regulation of rho- and rap-small GTPases signaling modules in the context of chemokine-triggered integrin activation (see JCB ref). We are investigating many signaling mechanisms involved in integrin activation, including analysis of different tyrosine kinases and phosphatases regulating the rho and rap modules on integrin activation. Systematic application of a combined approach of signal transduction functional interference, including chemical inhibitors, CPPs and siRNA, allows analyzing the signaling events regulating integrin-dependent leukocyte trafficking in murine and human primary cells and under physiological conditions, both in vitro and in vivo.
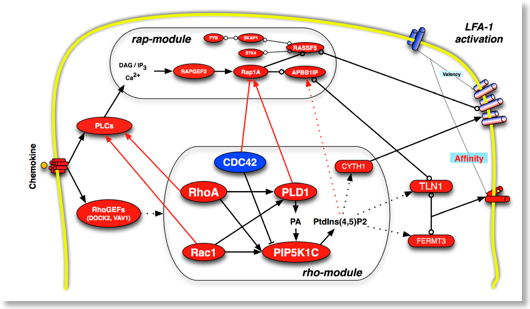
Montresor A, Toffali L, Constantin G, Laudanna C. Chemokines and the signaling modules regulating integrin affinity. Front Immunol. 2012 May 25;3:127. doi:10.3389/fimmu.2012.00127. eCollection 2012. PubMed PMID: 22654882; PubMed Central PMCID: PMC3360201.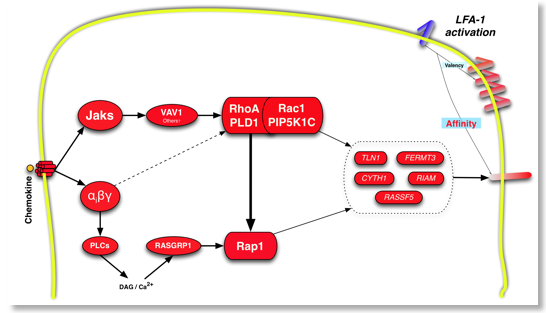
Montresor A, Bolomini-Vittori M, Toffali L, Rossi B, Constantin G, Laudanna C. JAK tyrosine kinases promote hierarchical activation of Rho and Rap modules of integrin activation. J Cell Biol. 2013 Dec 23;203(6):1003-19. doi: 10.1083/jcb. 01303067. PubMed PMID: 24368807.
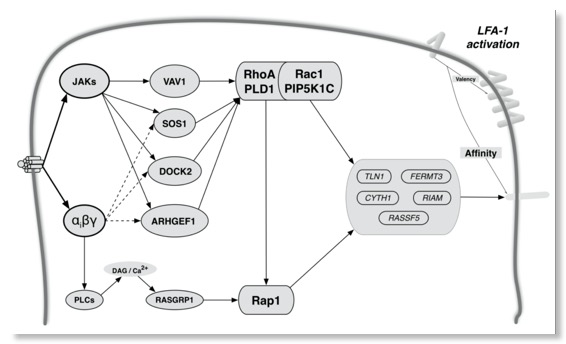
Toffali L, Montresor A, Mirenda M, Scita G, Laudanna C. SOS1, ARHGEF1, and DOCK2 rho-GEFs Mediate JAK-Dependent LFA-1 Activation by Chemokines.. J Immunol. 2017 Jan 15;198(2):708-717. PMID: 27986909
Role of protein tyrosine phosphatases in leukocyte trafficking (PTPs structure diagrams)
Signal transduction is regulated by the balanced activity of protein and lipid kinases and phosphatases, overall called the kino-phosphatome. The regulatory role of phosphatases is much less characterized with respect to that of kinases. Little is known about the role of protein phosphatases in the regulation of leukocyte trafficking. We are studying the role of receptor protein tyrosine phosphatases (PTPRs) in the regulation of leukocyte trafficking. Specifically, we are studying the role of PTPRG as potential regulator of chemoattractant-induced integrin activation in human primary monocytes and B-lymphocytes, either as modulator of signaling events as well as homing receptor.
We have developed a novel approach based on CPPs to selectively up-modulate the tyrosine phosphatase activity of PTPRG and infer its role in leukocyte trafficking. A Penetratin-1 (P1) based fusion peptide has been designed including the wedge domain (WD) identified in PTPRG. This P1-WD peptide specifically activates the tyrosine phosphatase activity of PTPRG. Moreover, TAT-PTPRG fusion CPPs have been generated, including the entire cytoplasmatic region of PTPRG, either wild-type (constitutive tyrosine phosphatase activity) as well as mutated (D1028A, without tyrosine phosphatase activity but phosphosite trapping capability) (see ![]() animation). These CPPs are tested in chemoattractant-induced leukocyte trafficking (Mirenda et al. JI 2015). The CPPs have been also used to preform a high throughput phosphoproteomics analysis in fMLP-treated human monocytes (see
animation). These CPPs are tested in chemoattractant-induced leukocyte trafficking (Mirenda et al. JI 2015). The CPPs have been also used to preform a high throughput phosphoproteomics analysis in fMLP-treated human monocytes (see ![]() Movie). We are also investigating the role of PTPRG as homing receptor.
Movie). We are also investigating the role of PTPRG as homing receptor.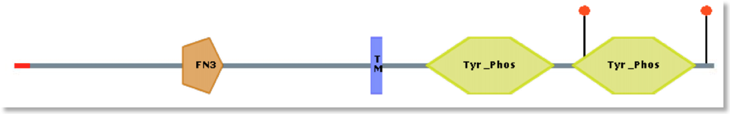 PTPRG domains
PTPRG domains 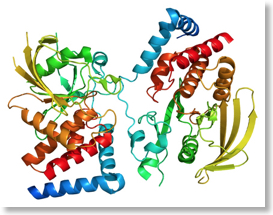 PTPRG 3D
PTPRG 3D
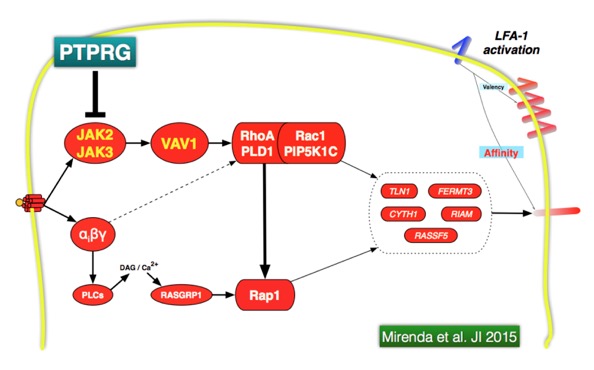
Mirenda M, Toffali L, Montresor A, Scardoni G, Sorio C and Laudanna. Protein tyrosine phosphatase, receptor type, gamma (PTPRG) is a JAK phosphatase and negatively regulates leukocyte integrin activation. Journal of Immunology 2015 Mar 1;194(5):2168-79 PubMed PMID: 25624455.
Molecular mechanisms of leukocyte deformability and mechano-transduction
Leukocytes spend the majority of their life flowing in the blood and lymphoid vessels. Here leukocytes experience consistent mechanical stress, either by vessel wall shear stress and by shape deformability induced by continuous entry and exit in and out the capillary nets (see picture below). The molecular mechanisms ensuring resistance to mechanical stress and allowing shape stability in spite of continuos deformation are still unknown. Moreover, the regulatory meaning of mechano-transduction and deformability is still unexplored. We have developed microfluidic devices and signaling interference approaches to investigate the molecular events regulating leukocyte deformability and its role in leukocyte homeostasis. 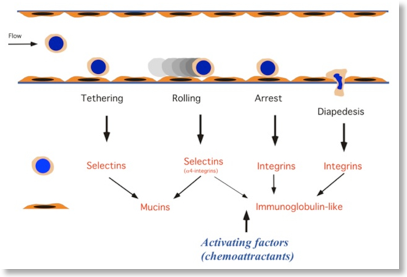 The step model of leukocyte recruitment
The step model of leukocyte recruitment 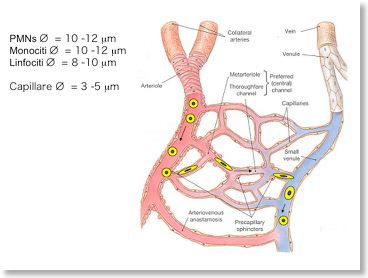 Deformability in capillary beds
Deformability in capillary beds
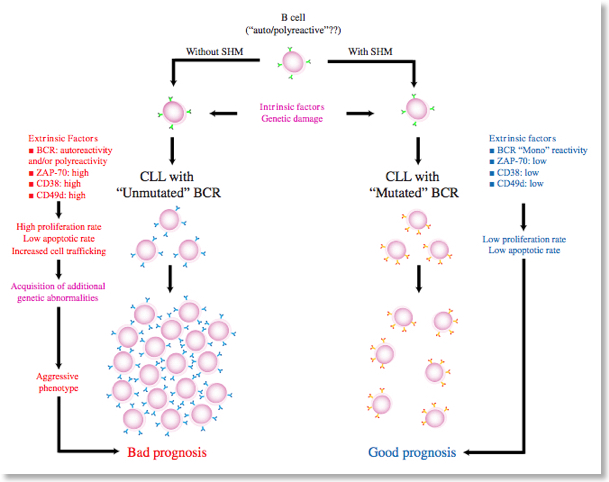
B-Cell chronic lymphocytic leukemia (B-CLL) pathogenesis (link1, link2)
B-cell chronic lymphocytic leukemia (B-CLL) is the most common leukemia in western countries, characterized by progressive accumulation of functionally incompetent B-lymphocytes in blood, bone marrow and secondary lymphoid organs. Abnormal signal transduction along with anomalous chemokine- and integrin-mediated trafficking of B-cells, determines permanence in bone marrow and accumulation in secondary lymphoid organs leading to cell substitution, immunodeficiency and resistance to therapy. B-CLL cells are characterized by loss of appropriate apoptosis, although this biologic feature is lost when B-CLL cells are removed from their environment, suggesting a critical role for micro environmental factors and/or adhesive interactions.
see B-CLL PubMed refs and molecular alterations in B-CLL
Regulation of B-CLL cell trafficking and survival (AIRC supported)
We have recently reported that, in B-CLL cells, the CXCL12-triggered rho-module of LFA-1 affinity modulation we recently described (Bolomini-Vittori et al., Nature Immunology) is functionally and biochemically altered (Montresor et al., Cancer Research). Overall, B-CLL cells experience an environment-dependent unbalance between pro- and anti-apoptotic signaling mechanisms, along with abnormalities of chemokine-induced inside-out signaling leading to altered mechanisms of integrin activation and adhesion. We have ongoing projects aiming to discovering the signal transduction characteristics and/or abnormalities controlling B-CLL lymphocyte trafficking to lymphoid organs. The project is carried-out in collaboration with Prof. Fabrizio Vinante and Dr. Antonella Rigo, Hematology division, Department of Medicine, University of Verona.
Phosphoproteomics analysis of B-CLL cells (AIRC supported)
Signal transduction abnormalities in B-CLL cells are also studied in the context of chemokine-triggered phospho-proteome. High-throughput analysis of phospho-proteome is performed by exploiting the Kinexus antibody-array service combined to mathematical procedures of network topological analysis in collaboration with the Center for Biomedical Computing (CBMC). B-lymphocytes isolated from several healthy donors or from CLL patients are systematically analyzed and computationally compared to identify CLL-specific signature of disease.
Metabolomics analysis of B-CLL cells (AIRC supported)
Metabolism is becoming increasingly important to comprehend cancer development and progression. The Warburg effect is a hallmark of cancer cells and its normalization is considered an important target for cancer therapy. We are pursuing projects to study metabolic alterations in B-CLL. Metabolic abnormalities in B-CLL cells are studied in the context of chemokine signaling. High-throughput analysis of the metabolome is performed by exploiting the Metabolon service associated to mathematical procedures of directed network topological analysis in collaboration with the Center for Biomedical Computing (CBMC). B-lymphocytes isolated from several healthy donors or from CLL patients are systematically analyzed and computationally compared to identify CLL-specific metabolic anomalies characterizing CLL.
Bioinformatics and topological analysis of biological networks in Systems Biology
High-throughput ‘omics technologies provide massive information about functional states of several molecules at once, thus greatly speeding up the discovery. However, they do represent a formidable challenge of interpretation of experimental data. Poor analysis of high-throughput data may hamper gaining a deeper knowledge on complex biological processes, preventing the identification of network signatures of disease. At the CBMC, we have developed a standard statistical and mathematical procedure to analyze high-throughput ‘omics data, specifically proteomics and metabolomics data. Raw data are normalized to background noise, filtered for higher significance and statistically clustered by means of a number of statistical methods, including analysis of co-variance, parametric and non-parametric tests, z-test and z-ratio computation (S. Zorzan). Once experimental data are filtered and the most significant data are extracted from the bulk of raw data, we proceed by applying a bioinformatics network analysis tools developed by the CBMC (G. Scardoni) in the context of annotated network topological analysis. Here, we pursue the determination of network structures centered on the identified protein and/or metabolites, to infer potential mechanisms of regulation. To this end, we apply a number of algorithms of network topological (static) analysis, with specific focus on topological cluster analysis, modular decomposition and node centrality analysis, altogether allowing the determination of network functional structures and modules and the categorization of most relevant regulatory nodes. Categorized and prioritized nodes and network structures are, then, the focus for deeper experimental analysis and verification. By iterating this procedure we progressively refine the analysis and identity new mechanisms of regulation (see refs.).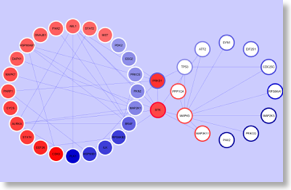 A phosphonetwork involved in B-CLL pathogenesis
A phosphonetwork involved in B-CLL pathogenesis
Trojan nanovectors (CPPs) in nanomedicine
Cell-penetrating peptides (CPPs), also called trojan nano vectors or trojan peptides, are carriers for soluble, high molecular weight, molecules that are normally unable to overstep the cell plasma membrane. Penetratin-1 (P1), from Antennapedia, and TAT, from HIV, are the best studied and characterized example of CPPs and their efficiency as systems for intracellular delivery of long peptides, entire proteins or nano particles has been widely tested for hundreds of cargos, including active proteins, and was demonstrated under several conditions, either in vitro as well as in vivo, and in different cellular contexts, including a variety of primary mammalian cells. CPPs allow the intracellular transfer of various cargoes, from small chemical molecules to nano-sized particles, siRNA and large fragments of DNA. CPPs biological effects show a dose-response curve; thus, CPPs do display a pharmacological profile. Because of this ability, CPPs hold great potential as in vitro and in vivo delivery vehicles for use in research and for the targeted delivery of therapeutics to individual cells. Indeed, P1 and TAT and related peptides are under extensive testing for developing novel pharmacologic approaches. Cell-penetrating peptides are able to enter cells by means of a number of mechanisms, including direct passive entry, micropinocytosis and active endocytosis, a process by which cells actively internalize molecules by engulfing them.
We have ongoing projects to validate the usage of CPPs as novel therapeutical agents capable of modulating critical signaling pathways in pathology such as inflammation, EAE, B-CLL and extracorporeal circulation (ECC) (in collaboration with prof. G. Faggian). The project is carried out in the context of the Nanomedicine project (WP4, coordinated by prof. L. Cominacini) (University of Verona), sponsored by Fondazione Cariverona. Go here for the Nanomedicine European Technology Platform (ETP).